|
The opportunity to experience Rare Disease Week in our nation's capital was more than I can describe in words. I was surrounded by families and patients walking the same walk and fighting many of the same battles. Some are still advocating even after the loss of a child, and many were patients themselves, dealing with life limiting/robbing condititions. The week started at the NIH, where researchers, policymakers, and advocates shared recent developments that are further advancing rare disease treatments. In 2016 alone, the NIH Clinical Center involved over 15,300 rare disease patients in a clinical trial or natural history protocol. The NIH's focus on rare disease research is intentional - it is often very easy to asemble a cohort of patients with rare diseases. But even more promising, rare diseases often provide a window to common diseases. Shared during the NIH Rare Disease Day was this quote from William Harvey in 1657: Nature is nowhere accustomed more openly to display her secret mysteries than in cases where she shows traces of her workings apart from the beaten path; nor is there any better way to advance the proper practice of medicine than to give our minds to the discovery of the usual law of nature, by careful investigation of cases of rarer forms of disease. The stage was set Tuesday for our legislative meetings though our Rare Disease Legislative Conference, sponsored by Rare Disease Legislative Advocates, and culminated in Presendent Trump's address to congress, where he honored a student with a rare disease and promised a nation of "miracles" by slashing restraints at the FDA and across the government. And I was thrilled to see my representative Congressman Bill Posey wearing the blue demin rare disease ribbon that we gave him the week prior. On Wednesday, 328 rare disease advocates participated in 270 Lobby Day meetings with US Senators, Congressmen, and staff. Advoates shared their personal stories, and asked that policymakers help the rare disease community (over 20 million Americans) with key issues. We asked for robust funding for the NIH and FDA, and to implement approapriations from the 21st Century Cures, which passes last year by 392-26 in the House and 94-5 in the Senate. We asked Congressman and Senators to support the OPEN ACT, which was pulled from 21st Century Cures at the last minute. The OPEN ACT would repurpose approved drugs for rare disease uses, and is estimated to double the number of drugs available to rare disease patients. This would have a significant imact on our community, as 95% of rare diseases have no FDA-approved treatment. And finally, we asked to ensure that rare diease patients don't lose access to affordable, life-saving heath insurance coverage in the upcoming policy changes. I thank our Florida Congressmen, Senators, and staff for taking the time to meet with us and hear from the rare disease community. Thank you to Congressman Posey for honoring Rare Disease Day with your ribbon and for participating in the Rare Disease Congressional Caucus. And we are especially grateful to Congressman Darren Soto for commiting to co-sponsor the OPEN ACT after meeting with our group. On Thursday, we visited the FDA White Oak campus to see Curren's Beyond the Diagnosis portrait, which has been on display along with 14 other paintings in honor of Rare Disease Day. We met with a wonderful woman from the office of Orphan Products Development and she shared the gallery with us. It was very overwhelming to see Curren's painting in person, and to experience the exhibit and the beautiful art created to honor these special children. After the FDA trip, we headed back to D.C. for the Rare Disease Congressional Caucus briefing on the topic of advancing rare disease treatments in the era of cures and health care reform. This was a wonderful recap of all the critical issues we focused on throughout the week, and it's wonderful to see that over 100 Senators and Congressmen are members. I left Washington D.C. feeling on top of the world, but in recent weeks I see that there is so much work to be done in the Rare Disease advocacy space. If you follow me on facebook, you may know that Curren has just lost his Children's Health Insurance Program (CHIP) coverage for medically complex children (CMS Title 21). If Medicaid is decentralized, block-granted, and/or per capita capped, it would be tragic for children with complex medical needs and developmental disabilities, as it covers nearly half of all children with special healthcare needs. There is not a private insurance plan currently available to me at this time that covers the therapy and equipment that my son needs. Additionally, it is very troubling to hear of the proposed cut of $6 billion dollars from NIH funding (almost 20% of the current budget). I hope that we are able to fund innovation to create a nation of "miracles" as referenced in the Joint Session on Rare Disease Day, and that we don't overlook our most vulnerable population of disabled and sick children in our years to come.
1 Comment
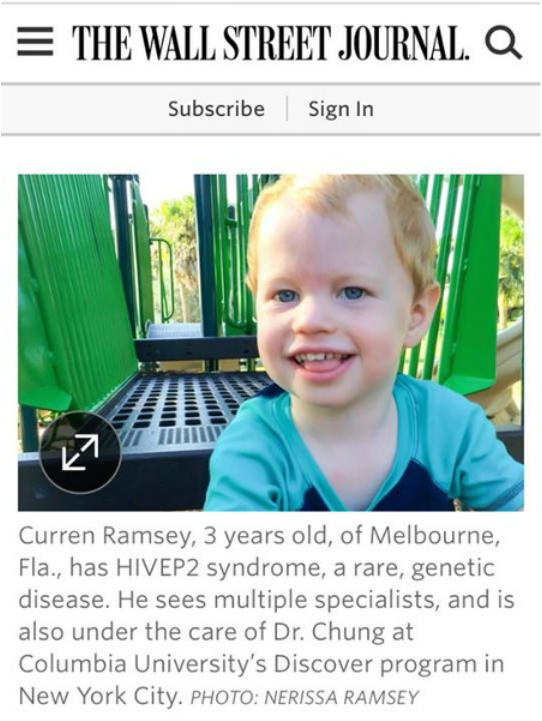
As I prepare for an incredible week in our nation's capitol - I can't help but get butterflies knowing that I am about to experience phenomenal opportunties to advocate for rare diseases at the National Institutes of Health, Capitol Hill, and the FDA. I am humbled to be able to meet with researchers, doctors, legislators, and scientists and share Curren's story along with other advocates from all over the country. While there are currently only 15 children known to have HIVEP2 disorder, one in 10 Americans battle a rare rare disease. We will be coming together as a community to share our voice on the critical issues our children face. I would like to share some words from the Rare Disease Day website on this year's theme - research: Imagine going to see your doctor only to be told that they don’t know what is happening to your body, that they don’t know what your disease is. Imagine that they can diagnose your disease but tell you that there is no cure or even treatment available. Or that the treatment available is not fully effective but just the best possible option. You don’t know how you or your loved one will manage life from one day to the next, nor how the disease will affect your work or school life. Imagine what it would be like to live without answers to your most basic questions. This is the reality for many rare disease patients. Research can lead to the identification of previously unknown diseases and can increase understanding of diseases. It can enable doctors to give a correct diagnosis and provides information to patients about their disease. It can lead to the development of new innovative treatments and in some cases a cure. Research is key. It brings hope to the millions of people living with a rare disease across the world and their families. Rare disease research is crucial to providing patients with the answers and solutions they need, whether it’s a treatment, cure or improved care. On 28 February 2017, the tenth edition of Rare Disease Day will see thousands of people from all over the world come together to advocate for more research on rare diseases. Over the last few decades, funds dedicated to rare disease research have increased. But it can’t stop there. Rare Disease Day 2017 is therefore an opportunity to call upon researchers, universities, students, companies, policy makers and clinicians to do more research and to make them aware of the importance of research for the rare disease community. Rare disease patients and families, patient organisations, politicians, carers, medical professionals, researchers and industry will come together to raise awareness of rare diseases through thousands of events all over the world. Rare Disease Day 2017 is also an opportunity to recognise the crucial role that patients play in research. Patient involvement in research has resulted in more research, which is better targeted to the needs of patients. Patients no longer solely reap the benefits of research; they are empowered and valued partners from the beginning to the end of the research process. Please join me in spreading the word for our HIVEP2 children and millions of others living with severe and lifelong conditions that have no treatments or cures. Alone we are rare, together we are strong! I have been honored with an incredible opportunity - to represent the rare disease community in Washington DC during the 2017 Rare Disease Week on Capitol Hill. Hosted by Rare Disease Legislative Advocates, hundreds of rare disease community members from across the country will join together to learn about federal legislative issues, meet other advocates, and share their unique stories with legislators. I am thrilled to be participating in this series of events, and very excited to share Curren's voice (and many others, too!) with our Congressmen and Senators. My goal is to raise legislative awareness for the needs of the rare disease community, and address roadblocks in the development and access of critical treatment and management options. There are three specific issues that I will be focusing on during my trip, which not only impact the rare disease community, but also much of the general American population. The first critical issue is the lack of treatment and management options. At three years old, Curren has seen over 20 doctors, and none of them have been able to prescribe any type of medication or treatment that has helped to manage any of Curren's symptoms. In fact, many of his symptoms are getting worse over time. This is not uncommon in the rare disease community. Statistics show that there are over 7000 rare diseases and less than 500 FDA-approved treatments for those rare diseases - that leaves 93% of rare diseases with no treatment or management options. Sadly, many rare diseases are terminal, and 30% of patients die before their 5th birthday, This skews the patient population so that over half of those battling rare diseases are children, There needs to be a stronger emphasis on orphan drug expansion, increasing FDA funding, and modernizing clinical trials to help those who are running out of time. The second critical issue is healthcare policy. Up until last year, Curren was on private insurance through my employer, During that time, we were denied coverage for speech therapy (for a completely non-verbal child), physical therapy (for a non-ambulatory child), occupation therapy (for a child who cannot feed himself), ABA therapy (for a child with autism), B12 and folinic acid supplements (because they aren't FDA approved), spio compression suits (because of medical coding issues), and whole exome sequencing (because it wasn't "medically necessary"). That's right - Curren's genetic test that actually provided his diagnosis was found to be not medically necessary, even after appeal, and was not covered under private insurance. This decision was on the premise that the small chance of "meaningful" information resulting was not worth the cost, and regardless should not change his plan of care. I do beg to differ. Now we know that Curren has a lifelong disease that is due to a mutation in a gene, he will not "grow out of it" as some doctors said early on, and it absolutely does change his plan of care. This past year, Curren was eligible for a state CHIP insurance program for medically complex children, and it has been a tremendous blessing, This program has covered all the therapies, adaptive equipment, and appointments that private insurance didn't. Because of our CHIP coverage, Curren has been able to go to the best pediatric rehabilitation facility in our county, get a wheelchair, see multiple specialists, get on a waiting list for ABA therapy, and much more, I am concerned that whether public or private, children with profound medical conditions and disabilities do not have access to the therapies and early interventions that will allow them to be their personal best in life. I know that healthcare is a very politically charged issue right now, and I don't want to get into politics in this post. I do, however, feel that whether conservative or liberal, we have a duty to protect our most vulnerable and maintain a system that allows children with or without complex needs the opportunity to thrive. My last critical issue is one that really applies to us all, and that is one of advancing research. Where would we be without science? W. Edwards Deming once said, "without data you're just another person with an opinion." Whether in my professional life as an architect, or in a personal capacity as an advocate, data is needed to create a defendable foundation. Now more than ever, we should be removing barriers to increase research collaboration and investing in 21st century science, I have to say, I was not happy to see potential FDA commissioner Joseph Gulfos's position in his recent op ed, stating that he wants to see more research funding shifted to supporting treatments for obesity, diabetes, and cardiovascular disease, and away from rare diseases. I would first like to point out that rare disease research helps us understand medicine for all diseases. One of many examples being this story of a rare lysosomal storage disorder informing therapeutic targets for Parkinson's disease. Secondly (and selfishly), I am a bit offended about the idea that we would give up on diseases that primarily affect children that have no treatment or management options to focus on often lifestyle-related conditions that already have FDA-approved treatments. I am not trying to stir controversy, but I would be thrilled to have an option of surgery or medication or medical device or dietary change that would improve my son's quality of life - but unfortunately none of these are an option to me. I will be a squeaky wheel, as Joseph Gulfo points out, until my son has options available to him to make his life better. To give him the opportunity to speak words (and anyone that knows Curren, knows he has a multitude of words to say) . To give him the opportunity to walk (or run, or chase his brother), To give him to them opportunity to not be a "failure to thrive". Please stand with us on February 28th, to show solidarity for the rare community. Alone we are rare, but together we are strong. So I am weeks late with this post, but a lot has happened in the past month - a new president-elect, a new baby nephew, the thanksgiving holiday, and the first HIVEP2 family conference (more on that later!). But on November 8th, our incredible doctor in New York was featured in the Wall Street Journal. As part of the article, Curren and HIVEP disorder were mentioned! Dr Chung has provided motivation for me to continue to fight for Curren and HIVEP2 advocacy. Between the research opportunities and family conferences, we have so many more resources available to us than what you would expect for a community of only twelve patients. Rare disease patients can often feel isolated, but the opportunities that Dr Chung provides our community gives me hope for the future, and they have the potential to change my son's trajectory in life. We are so honored and blessed to be under Dr Chung and her team's care, and are beyond thrilled to have HIVEP2 mentioned in a national newspaper. From the Wall Street Journal on November 8th, 2016, "The Doctors who Solve Medical Mysteries":
Our HIVEP2 community also had our first virtual family conference this past week, and it was so encouraging to connect with these families and researchers. I cannot say enough amazing things about the Simons VIP project and the work they are doing to further advance our understanding of the genetic components underlying autism and developmental disabilities. We can't wait to see how our community will grow in 2017. In other exciting news, the revised 21st Century Cures Act passed by a sweeping 392-26 vote in the House of Representatives last week! While not everything that would have hugely benefited the rare disease community was included (such as the OPEN Act, additional resources for developing natural history registries for rare diseases, and more robust funding for the NIH and FDA), the revised bill is still a great step forward for the development of therapies for rare diseases. The White House is urging the Senate to promptly pass this bill so that the President can sign it before the end of the year. Thank you all for the support I receiving following my last blog post on the 21st Century Cures Act. Most exciting of all, Curren has been getting used to his new wheelchair over the past month. Within the past few days, it has all come together and he is able to push his chair all by himself. This is short of incredible, as many things that most find easy can be incredibly challenging for Curren. After 3 years, my little guy is finally able to move around without relying on others, and it is the greatest feeling in the world! With the holidays coming up around the corner, we have been talking a lot about Christmas lists. My 7 year old son Weston already knows precisely what he wants: a play station, a bow and arrow, and some magic tricks. Since Curren isn‘t yet able to talk, I told Weston he has the important task of coming up with the perfect list for Curren, and telling Santa what his brother wants. We have been running through lots of ideas, but what do you get a sweet 3-year old with a rare neurodevelopmental disorder who would rather high-five everyone in the room than play with toys? There are tons of therapy tools and adaptive equipment that would be helpful for Curren, but that’s really not all that fun for a little guy that already does 9 hours of therapy every week. I asked Weston what he thought the one thing in the world his brother would want for Christmas more than anything else would be, and his answer was difficult - he would want to walk and talk and play with friends. That really would be the ultimate gift, but how in the world do we get there? The majority of my time and thoughts have been consumed with this idea for over a year now. It’s the reason for the trips to New York and Baltimore, for my husband leaving his job, for the challenging gluten-free diary-free diet, for the midnight googling and worrying, for the pubmed alerts, for the intensive therapies, for the B12 injections - all for the hope that we stumble upon a doctor or researcher or therapy than can level the playing field a bit more and help Curren to achieve his personal best in life. How do we get more research, better management, more treatments, and eventually a cure? We are not the only family. It is estimated that over 30 million Americans are battling a rare disease today, and half of those affected are children. Of those children, 30% will not live to their 5th birthday. Over half of all rare diseases don’t have a specific foundation supporting or researching their condition (including Curren's condition, HIVEP2 disorder). How do we get cures now? There is hope today for the future. For my family and millions of others, it is found in the 21st Century Cures Act, which passed 343-77 in the House of Representatives last year, but has not yet passed through the Senate. Chairman Fred Upton (R-Michigan) calls the landmark innovative effort a “once-in-a-generation, transformational opportunity to change the way we treat disease.” Senate Majority Leader Mitch McConnell (R-Kentucky) said that the bill “could end up being the most significant piece of legislation we pass in the whole Congress." This act would not only benefit the rare disease community, but "could really change the face of cancer" according to Jon Retzlaff, managing director of science policy and government relations at the American Association for Cancer Research. If the 21st Century Cures Act passes, what would it mean for Curren, for others living with a rare disease, and for those battling cancer? it will accelerate the discovery, development, and delivery of life saving and life improving therapies, and transform the quest for faster cures. This will be achieved by removing barriers for research collaboration, by incorporating the patient perspective into the drug development and regulatory review process, by measuring success and identifying diseases earlier through personalized medicine, by providing new incentives for the development of drugs for rare diseases, and by investing in 21st century science and next generation investigators - all while keeping and creating jobs here in the United States. This act could double the number of therapies available for rare disease patients. This bipartisan act has been years in the making and is supported by hundreds of groups across the patient and research spectrum., but is in danger of not becoming law. There is word that some in Congress are attempting to remove substantial portions of the bill, including the Open Act, which would effectively kill the bill in Congress. A delay in legislation is a delay in life-saving treatment for patients. If you feel so inclined, please help us make cures a reality for Curren and millions of others! The #CuresNOW page provides many easy ways to promote the 21st Century Cures Act. Please help all those who want nothing more than cures for their loved ones this holiday! |
SignupSign up for our mailing list and you'll be the first to get updates on Curren and all his adventures!
About MeI am a mother, architect, wife, and a lover (not a fighter) - with a thirst for knowledge. My journey been recently refocused, as my family navigates through the world of medical and developmental uncertainty in hopes of providing every opportunity for my son to be his personal best in life. Categories
All
Archives
October 2017
|

















 RSS Feed
RSS Feed
